Development and Disease Control
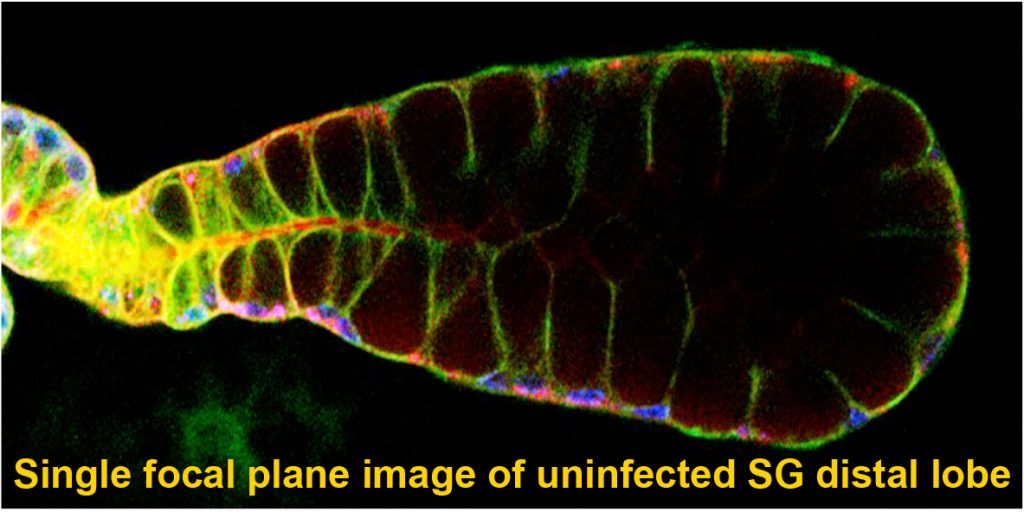
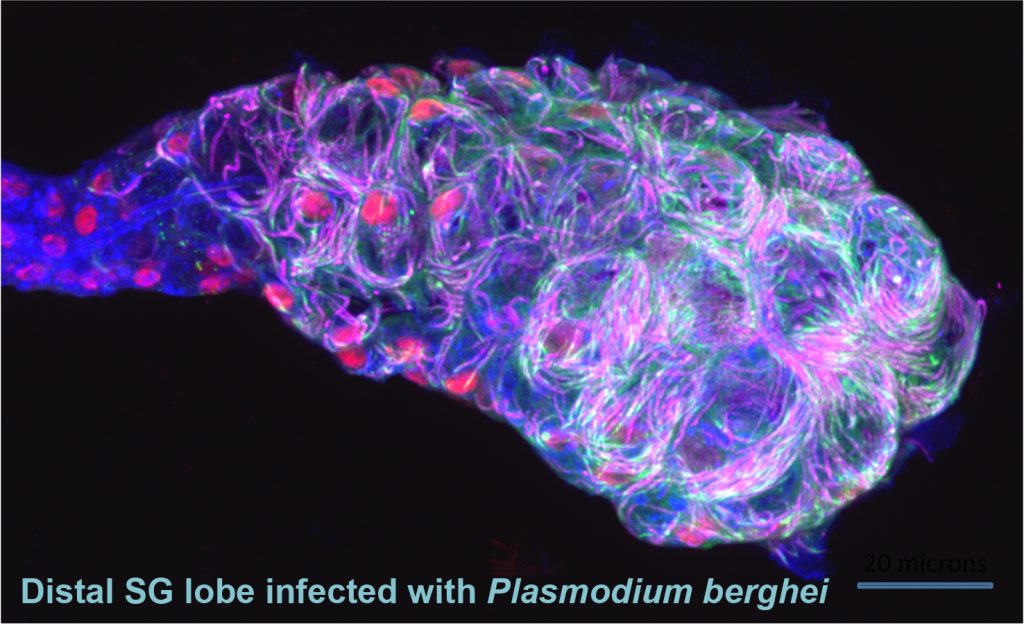
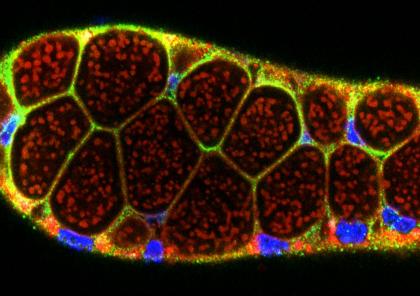
Mosquitoes and other insects are vectors for malaria, leishmaniasis, lyme disease, sleeping sickness, river blindness, Dengue fever and Zika – diseases that affect hundreds of millions of people and cause several million deaths every year. Most of these diseases require the disease-causing pathogen to traverse the insect salivary gland (SG) for transmission to humans. Despite the importance of the SG in the life cycle of insect-borne pathogens, almost nothing is known about the factors that drive the formation, function, and homeostasis of SGs in any insect outside of Drosophila. Our long-term goal is to develop and test strategies to block parasite transmission by intervening at the level of the mosquito vector, preventing transmission of disease by causing the timely death of the mosquito SG. Our studies leverage the considerable expertise of the Andrew lab regarding Drosophila SG biology to learn how mosquito SGs form and are maintained. Our initial goal is to determine if the mosquito versions of key transcription factors that function in Drosophila salivary gland specialization, survival and morphogenesis play the same critical roles in the adult mosquito SG. If so, our long-term goal is to target these genes in a tissue and time-specific manner as an additional strategy for limiting transmission of malaria. Importantly, this strategy should be generally applicable to preventing transmission of many insect borne human diseases.