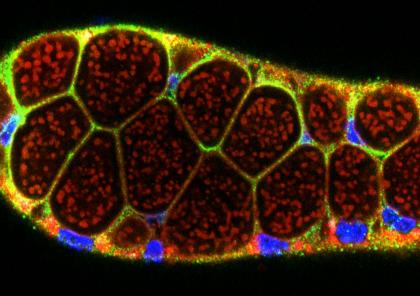
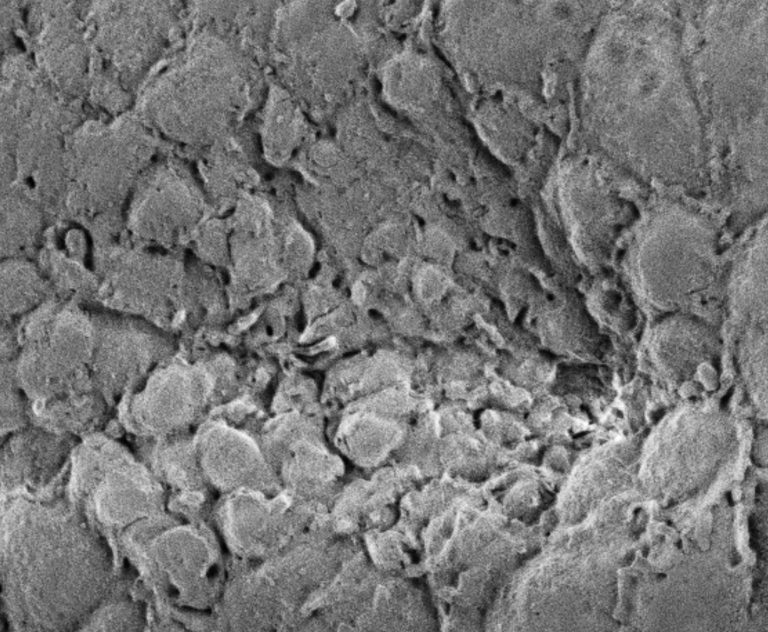
The Andrew Lab studies how organs are specified, form and specialize using the Drosophila salivary gland and trachea as model systems. We, and others, have identified the factors required to specify salivary gland and tracheal cell fates. Our lab has also characterized many of the earliest-acting tissue-specific transcription factors. These transcription factors maintain specialized cell fates, control morphogenesis, and activate tissue-specific gene expression patterns. In both the salivary gland and trachea, we have discovered many downstream targets of the early-expressed tissue-specific transcription factors. The functions of these target genes have revealed an unexpected parsing of cellular functions to different tissue-specific transcription factors and many of these functions are conserved among family members in higher eukaryotes. We continue to study the contributions of key downstream targets in both tissue specialization and the morphological transformation of tissue primordia into mature functional architectures. We are now also characterizing adult mosquito salivary glands from two human vector species as the first steps toward leveraging our findings on the Drosophila salivary gland to develop strategies to limit transmission of malaria and other mosquito-borne diseases.
The Andrew Lab uses a combination of genome-wide DNA binding and gene expression studies, classical and modern genetic and molecular biological approaches, as well as advanced microscopy and image analysis to uncover the gene networks governing organ development.