Organ Specification and Homeostasis
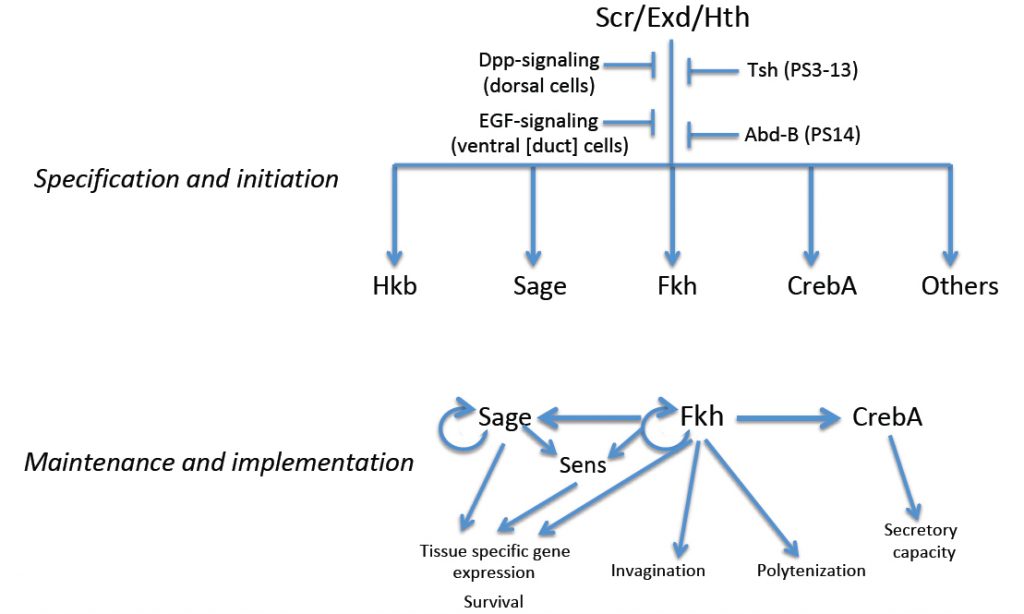
Drosophila salivary gland (SG) formation requires the Hox transcription factors, Sex combs reduced (Scr), Extradenticle (Exd) and Homothorax (Hth); in loss-of-function mutants for any of the corresponding genes, SGs fail to form. Moreover, ectopic expression of Scr, the one component of the complex with limited spatial expression, leads to the formation of additional SGs. Expression of Scr and hth and nuclear localization of Exd disappear, however, shortly after the SG initiates morphogenesis, suggesting that genes that specify the Drosophila SG cell fate are not involved directly in its terminal differentiation. Genes encoding several transcription factors are expressed in the early SG under the control of Scr, Exd and Hth, including fork head (fkh), which encodes a winged-helix transcription factor, CrebA, which encodes the Cyclic AMP Response Element Binding protein A, sage, which encodes an SG-specific bHLH protein, and huckebein (hkb), which encodes an Sp1/egr-like transcription factor. A subset of these early expressed SG transcription factors (Fkh, CrebA and Sage) maintain their own expression and/or the expression of each other. Thus, the early-expressed SG transcription factors both maintain and implement the SG cell fate decision, with Fkh playing a major role in this process.
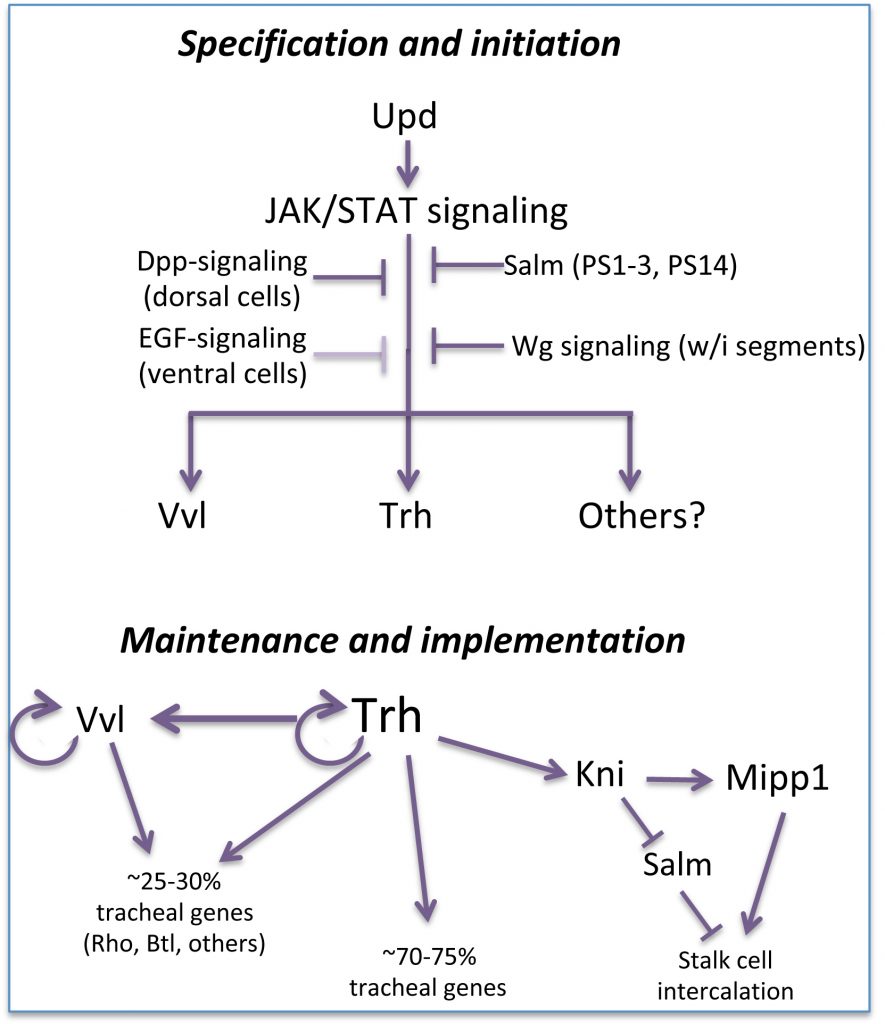
Drosophila embryonic trachea formation requires activation of the JAK/STAT signaling pathway by the Upd ligand. As with the SG, several factors, including the transcription factor Spalt as well as Wg-, Dpp- and EGF-signaling, limit trachea to only a subset of cells that experience JAK/STAT activation. Two major transcription factors are activated by the trachea-specifying genes: trachealess (trh) and ventral veinless (vvl). Both play major roles in maintaining and implementing the tracheal cell fate. Trh maintains its own expression and that of Vvl, and is required for expression of every known tracheal gene. Vvl is required for expression of an estimated 25-30% of tracheal genes. Thus, similar to the SG, multiple (at least two) transcription factors are required to both maintain and implement the tracheal cell fate decision, with Trh playing the major role in this process.